Soft corals of the Great Barrier Reef

Soft corals, sea fans and gorgonians are common names for a group with the scientific name Octocorallia or Alcyonacea. About 100 genera in 23 families are known to occur in shallow Indo-Pacific coral reefs. Octocorals are ecologically important components of the coral reef landscape, and being beautiful and colourful, are an attraction for divers.
Biology of octocorals
Octocorals can be distinguished from hard corals by the fact that octocoral polyps always have eight tentacles (Fig. 1), while hard coral polyps have multiples of six tentacles (hence their technical name 'hexacoral'). While the term soft coral is and sea fans are used for different growth forms (compact versus fan shaped), the differentiation between these groups has little scientific merit as their growth forms do not indicate taxonomic relationships, and many intermediate growth forms exist. The term octocoral' is therefore more appropriate.
Most soft corals lack a hard external skeleton, instead soft corals contain small calcareous sclerites in their body, which are used to identify the genera (Fabricius and Alderslade 2001) or species (through specialist taxonomic literature). A few species, such as the the blue coral (Heliopora) or the red organ-pipe coral (Tubipora) have a solid skeleton, superficially resembling that of hard corals. Despite their lack of reef-building capacity, octocorals are an important and diverse part of reefal and inter-reefal ecosystems.
Relatively few detailed octocoral studies exist (e.g., from the Great Barrier Reef and other parts of Australia, New Guinea, New Caledonia, Micronesia, Japan, South-Eastern Africa and the Red Sea), and there are still major gaps in the understanding of octocoral biogeography. Furthermore, the number of shallow-water Indo-Pacific octocoral species is unknown, as many species still await taxonomic description and many genera are in urgent need for revision.
Octocorals of the GBR include species with diverse biological attributes and contrasting ecological requirements. Many species are long-lived and slow-growing, while others are fast colonisers with a short life expectancy. Some large Sinularia colonies (family Alcyoniidae) are probably hundreds of years old, and some of the large gorgonian colonies may also be many decades old. Life expectancy and growth rates of most soft corals and gorgonians are however largely unknown, and as colonies may shrink when torn by storm waves or damaged by moving rubble or predation, there is a weak relationship between size and age.
Dispersal strategies also greatly vary among species, and include asexual propagation and sexual reproduction. Rapid colonisation of small patches of substratum often includes 'runner formation, budding of miniature colonies that fall of the mother colony and settle near-by, and fragmentation. Larger patches of bare substratum tend to be colonised by the settlement of sexually produced pelagic larvae. ‘Broadcaster’ taxa release their male and female gametes into the water column where they are fertilised; the pelagic larvae are dispersed by currents until they settle days to weeks later. ‘Brooder’ species have their eggs fertilised within the mother colony, and the resulting larvae develop on the colony surface until they detach and settle near the mother colony some days later.
In part, the food of octocorals consists of small suspended plankton particles filtered from the water column; they rely on water currents to carry the particles towards their polyps. Actively swimming zooplankton are not ingested. Much of the food is therefore derived from phytoplankton, minute detrital particles, flagellates and very small zooplankton. In addition, more than half of the octocoral taxa that grow in warm and shallow waters of the Indo-Pacific also contain endosymbiotic dinoflagellate algae (often called zooxanthellae) in their tissue, which fix carbon through photosynthesise and hence supply energy to their host (Fig. 2). The coral in return provides nutrients and shelter to the algae. This finely tuned symbiosis between animal and alga depends on the availability of light for photosynthesis. Symbiotic taxa (also called zooxanthellate or phototrophic taxa) include many representatives within the “true” soft corals (the Alcyoniina group), especially many of the genera within the abundant families Nephtheidae, Alcyoniidae and Xeniidae, but also members of most of the other large octocoral groupings. In contrast, most sea fans, and also several of the soft corals (e.g., Dendronephthya) do not contain dinoflagellate endosymbionts. These asymbiotic (azooxanthellate or heterotrophic) taxa are suspension feeders that strongly depend on currents to transport food particles towards the polyps; they are mostly found in high-flow environments. Heterotrophic taxa are easily visually distinguished from their phototrophic relatives by their bright yellow, orange, red, pink, purple or snow-white colouration (Fig. 2).
Ecology of octocorals
As sessile organisms without a protective skeleton, octocorals would appear to be vulnerable to predation. However with the exception of a few snails (e.g., the cowry shell Ovula ovum), fish (e.g., a few species of butterfly fish that selectively feed on coral and octocoral polyps) and the odd or accidental grazing by an echinoderm such as Diadema sea urchin, remarkably few organisms are able to feed on octocorals, and overall feeding pressure appears low. Many species are protected against predation, fouling by algae or overgrowth by neighbouring organisms through feeding-deterrent, toxic or allelopathic secondary metabolites. Many of these substances have been investigated for their bioactivity, and some may one day become pharmaceutically relevant. Octocoral colonies, although not contributing to reef growth, nevertheless provide shelter to a range of other reef inhabiting organisms. For example, some species of brittle star (Ophiuroidae), feather star (Crinoidea), shrimps, ctenophores and fish (gobies and pygmy sea horses) are exclusively found living on the surface of specific octocoral colonies. Most of these associates use the octocoral colony exclusively as perch or for shelter; however a few of these associates appear to also feed on the mucus of the octocorals.
After hard corals, octocorals are the second-most common group of macrobenthic animals on the GBR. Mean octocoral cover of the GBR regions ranges from 3% to 35% on outer reef slopes, but cover can be as high as 70% in current-swept yet wave-protected environments such as channels between reefs or islands, and near zero on wave-exposed macro-algal dominated turbid and silty inshore reef crests.
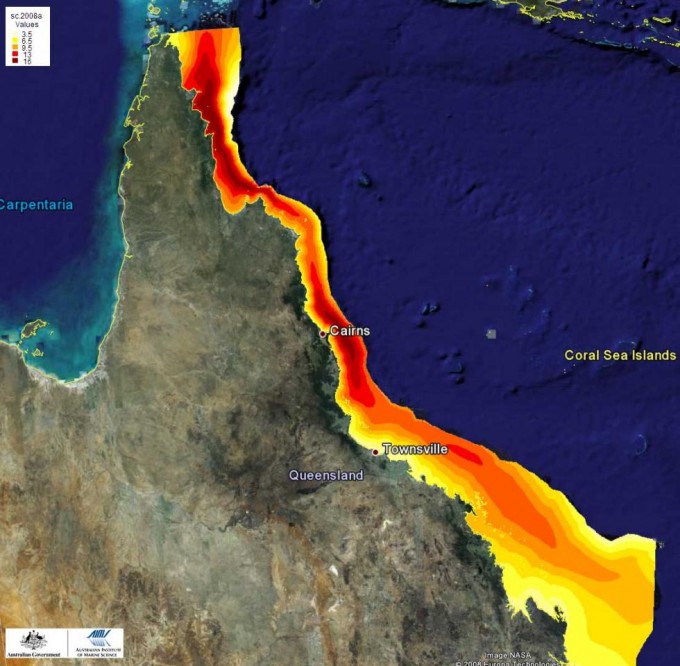
Octocorals are highly diverse not only taxonomically but also ecologically, including species with widely contrasting ecological niches and life history strategies. Richness increases from south to north (Fig. 3), and midshelf reefs have higher species richness than inshore and offshore reefs on the Great Barrier Reef (Fig. 3). Outer-shelf reefs contain diverse octocoral communities that are often characterised by high abundances of members of the family Xeniidae. Deep-water reef slopes and inter-reefal habitats are inhabited by azooxanthellate taxa such as many gorgonians and Dendronephthya, as well as ubiquitous and tolerant taxa such as Sinularia and Sarcophyton. Inshore reefs with fluctuating salinity and water clarity contain some species that are rarely found in clear-water habitats (e.g., Sinularia flexibilis, several species of Briareum and Solenocaulon), as well as a subset of those genera found in the clear-water habitats. Many of the Xeniidae and Nephtheidae are missing on turbid inshore reefs.
Ecological surveys have shown that the taxonomic richness of octocorals also increases from shallow water to moderate depths. It is further related to water clarity and amounts of sediments deposited. Changes in taxonomic richness and community composition in octocorals have therefore been suggested to be suitable as indicators of past and recent disturbance by poor water quality on the Great Barrier Reef and other reef environments.
Species richness of a given site is affected by three factors:
- First, the biogeographic location and colonization history of a region determines the regional species pool present. On the GBR, the species richness in octocorals strongly attenuates with increasing latitude: many more genera and species occur in the tropical far northern part than on the southern end of the GBR.
- Second, environmental conditions determine what cross-section of the local species pool occurs at that locality. In octocorals, abundances of particular taxa are strongly determined by the physical environment, especially turbidity, light availability and water currents.
- Third, at any point in time local and regional species richness also depend on disturbance history, specifically the nature and intensity of the disturbance, and the time since past disturbances have removed colonies. For octocorals, disturbances include storms with high wave energy (dislodging or damaging colonies), episodes of high water temperatures (causing coral bleaching), chronically reduced water clarity (reducing photosynthesis) and sedimentation (smothering colonies or hampering larval settlement). After a disturbance, the speed and efficiency of recolonization of a taxon will determine whether the taxon will again be present or not: fast colonizers continuously re-establish if propagules from surviving colonies (locally or further upstream) are available, whereas slow-colonizing or slow-growing taxa are unable to quickly return to their previous abundance. Similarly, chronic disturbance such as water pollution reduces diversity, because only persistent species can survive and flourish. In order to understand regional and local biodiversity patterns, biogeographic settings, environmental requirements of taxa and consequences of disturbances need to be investigated simultaneously.
References
Fabricius KE & Alderslade P (2001) Soft Corals and Sea Fans: A comprehensive guide to the tropical shallow water genera of the central-west Pacific, the Indian Ocean and the Red Sea. Australian Institute of Marine Science, Townsville. 264 pp. ISBN: 0 642 322104.
van Oppen MJH, Mieog J, Sanchez CA, Fabricius KE (2005) Diversity of algal endosymbionts (zooxanthellae) in octocorals: the roles of geography and host relationships. Molecular Ecology 14: 2403-2417.
Fabricius KE, Alderslade P, Williams GC, Colin PL, Golbuu Y (2007) Octocorallia in Palau, Micronesia: Effects of biogeography and coastal influences on local and regional biodiversity. Pages 79 - 91 in Kayanne H, Omori M, Fabricius K, Verheij E, Colin P, Golbuu Y, Yurihira H (eds) Coral Reefs of Palau. Palau International Coral Reef Centre.
Alderslade P, Fabricius KE (2008) Octocorals of the Great Barrier Reef. Chapter 21 in: Hutchings P, Kingsford M, Hough-Guldberg O (eds). The Great Barrier Reef. CSIRO Publishing, pp 222 - 245.
Fabricius KE and De'ath G (2008) Photosynthetic symbionts and energy supply determine octocoral biodiversity in coral reefs. Ecology 89: 3163 - 3173.